Langmuir 28, 15566-15576 (2012)
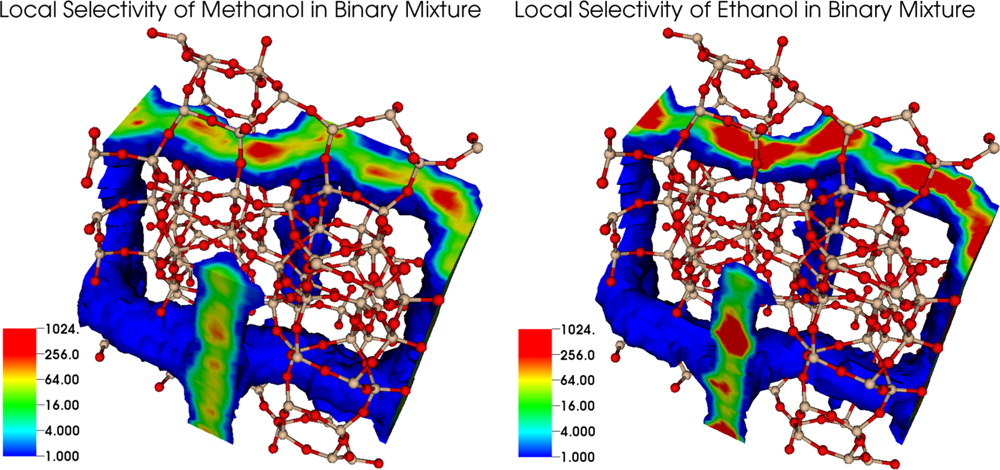
Configurational-bias Monte Carlo (CBMC) simulations in the isobaric–isothermal version of the Gibbs ensemble (GE) were carried out to probe the adsorption from aqueous solutions of methanol and/or ethanol onto silicalite-1. This methodology does require neither specification of the chemical potential nor any reference to activity models based on experimental data. The CBMC-GE methodology can be applied to the complete range of mixture compositions from pure water to pure alcohols and can also be used when multiple solute types are present at high concentration. The simulations demonstrate high selectivities for the alcohols (αethanol > αmethanol) almost over the entire composition range. The ideal adsorbed solution theory is found to substantially underpredict the amount of sorbed water and leads to very large errors for low alcohol solution concentrations. The simulations indicate that, at lower loadings, the adsorbed alcohol molecules can serve as seeds for water adsorption but, at higher loadings, alcohols displace water molecules from their preferred region.