J. Phys. Chem. B 102, 2578-2586 (1998)
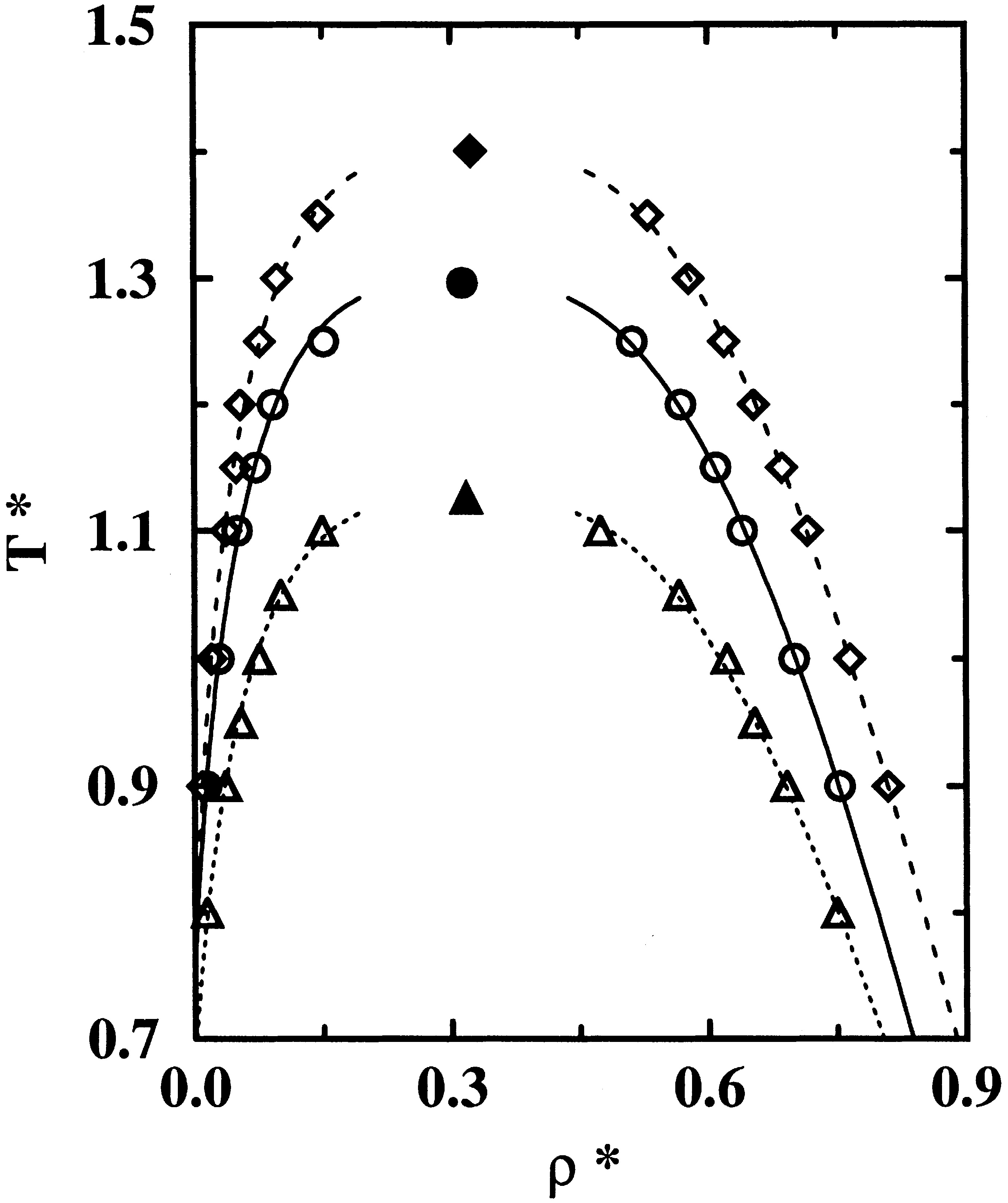
The performance of several all-atom force fields for alkanes is compared and evaluated. Configurational-bias Monte Carlo simulations in the Gibbs ensemble were carried out to calculate the vapor−liquid phase equilibria for methane, ethane, n-butane, n-pentane, and n-octane. The Williams, OPLS-AA, and MMFF94 force fields were selected as representative all-atom models for this study because they were fitted using three different strategies (Williams, crystal structures and heats of sublimation; OPLS-AA, liquid densities and heats of vaporization; MMFF94, rare gas pair potentials and quantum mechanics) and employ potentials with three different functional forms to describe nonbonded van der Waals interactions (Williams, Buckingham exp−r-6; OPLS-AA, Lennard-Jones 12−6; MMFF94, buffered 14−7). It is shown that seemingly small differences in the potential functions can account for very large changes in the fluid-phase behavior. The Williams and OPLS-AA force fields yield liquid densities, boiling temperatures, and critical points that are in acceptable, albeit not in quantitative agreement with experiments, whereas the fluid-phase behavior of the MMFF94 model shows very large deviations.