Sci. Adv. 4, art. no. eaao6283/11 pages (2018)
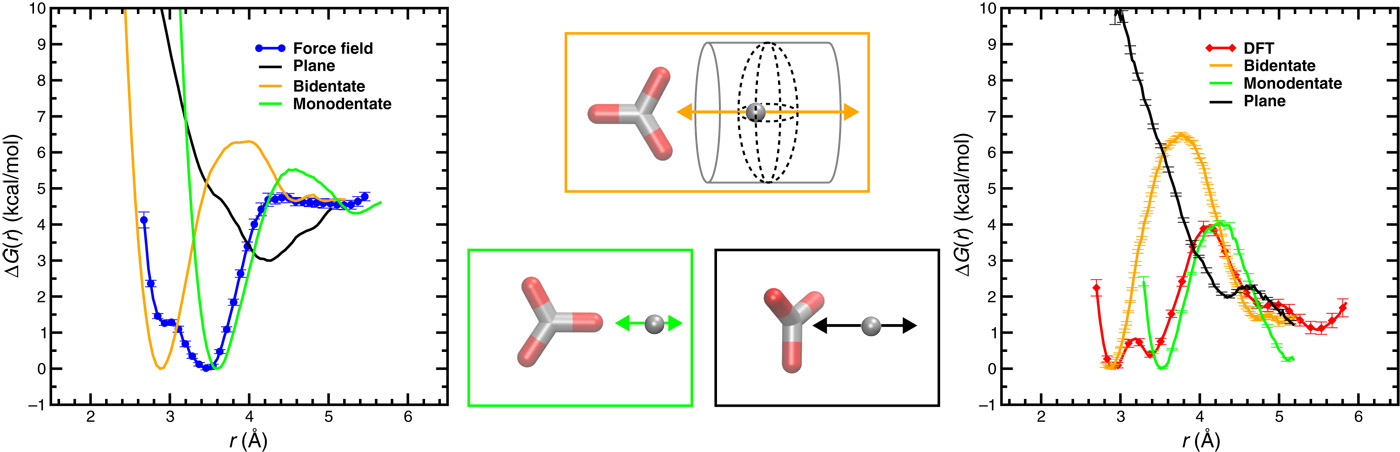
Mechanisms of CaCO3 nucleation from solutions that depend on multistage pathways and the existence of species far more complex than simple ions or ion pairs have recently been proposed. Herein, we provide a tightly coupled theoretical and experimental study on the pathways that precede the initial stages of CaCO3 nucleation. Starting from molecular simulations, we succeed in correctly predicting bulk thermodynamic quantities and experimental data, including equilibrium constants, titration curves, and detailed x-ray absorption spectra taken from the supersaturated CaCO3 solutions. The picture that emerges is in complete agreement with classical views of cluster populations in which ions and ion pairs dominate, with the concomitant free energy landscapes following classical nucleation theory.