J. Am. Chem. Soc. 124, 12232-12237 (2002)
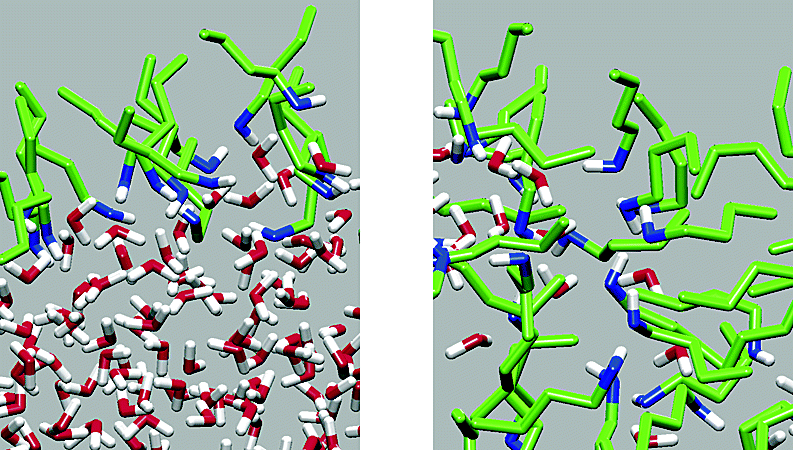
Adsorption and ordering at the vapor−liquid interfaces of mutually saturated water/1-butanol solutions at a temperature of 298.15 K were investigated using configurational-bias Monte Carlo simulations in the Gibbs ensemble and compared to the surface properties of neat water and 1-butanol liquids. A dense 1-butanol monolayer is observed at the surface of the water-rich phase, which results in a substantial decrease of its surface tension. In contrast, there is no enrichment of water molecules at the surface of the butanol-rich phase, and its surface tension is not significantly changed. Analysis of the interfacial structures reveals that these systems exhibit orientational ordering and composition heterogeneity. Analysis of the hydrogen-bonding distributions suggests that the formation of the 1-butanol monolayer is driven by an excellent match between water and the primary alcohol; that is, additional hydrogen bonds are formed between the excess free hydrogens of surface water and the excess hydrogen-bond acceptor sites of 1-butanol.